Story at a glance…
-
Within cancer tumors, fats build up in their oxidized form.
-
“Killer” T cells entering the tumor can become dysfunctional when feeding on oxidized fat cells, preventing them from targeting the cancer.
-
Oxidized fat occurs primarily through omega-six polyunsaturated fats like linoleic acid, found in vegetable and seed oils.
Researchers in the burgeoning field of immunometabolism have found that tumors have a natural metabolic-based defense mechanism against one of our body’s principal anti-cancer agents, the killer T cell.
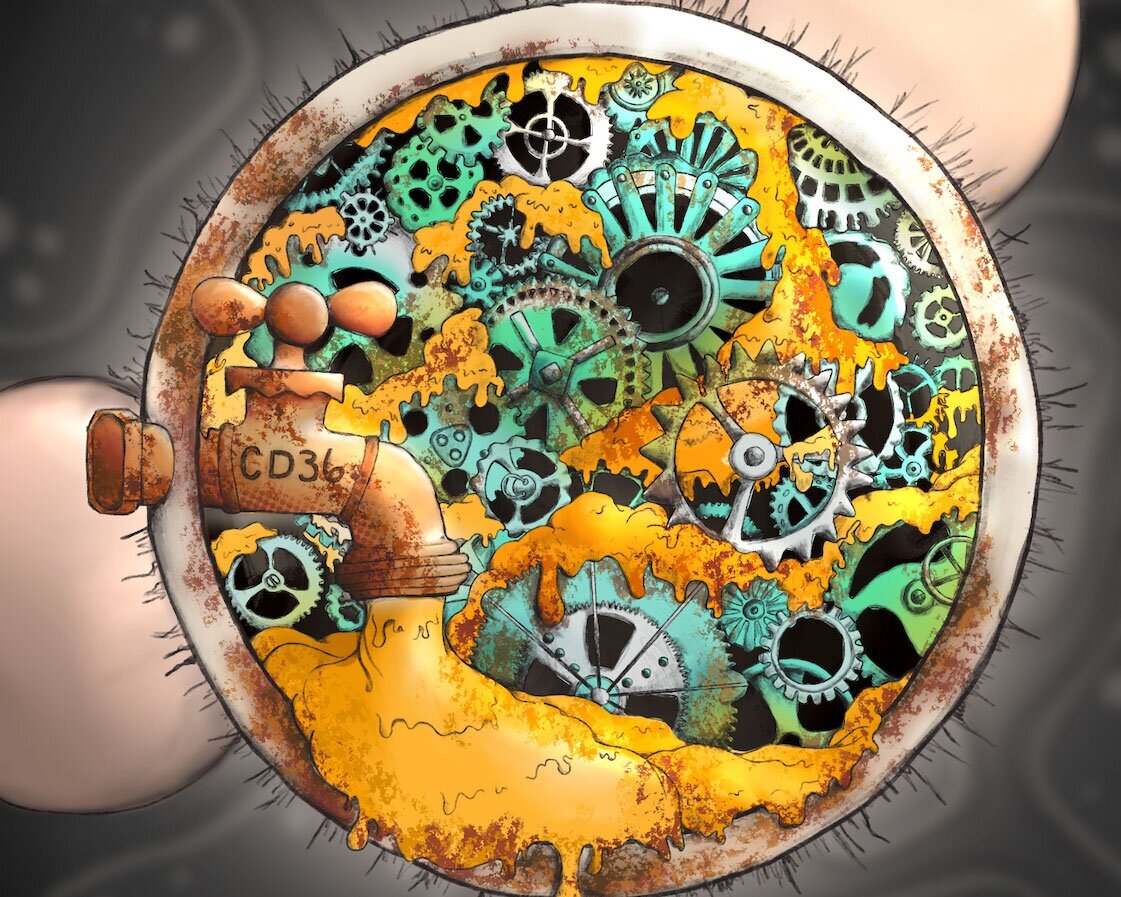
They found that as the killer T cell enters the tumor, the presence of oxidized fatty acids, like the ones derived in high amounts from industrial seed and vegetable oils, slow down and even impair the cell’s function. As the T cell ingests the oxidized fat it becomes starved of energy, and begins to make more of a fat energy transporter protein called CD36, which in a vicious cycle actually increases the amount and speed of its consumption of the oxidized fat.
In a paper published in Cell scientists at the Salk Institute for Biological Studies in California collaborated with others from Yale to produce a series of findings explaining this relationship between oxidized lipids (fats), cancer, and killer T cells for the first time, while simultaneously opening up a new avenue of immuno-metabolic therapies for cancer treatment.
“We know that tumors are a metabolically hostile environment for healthy cells, but elucidating which metabolic processes are altered and how this suppresses immune cell function is an important area of cancer research that is gaining a lot of attention,” says Susan Kaech, senior author and director of Salk’s NOMIS Center for Immunobiology, in a press release.
The first step to their conclusion was to establish that cancer tumors are indeed rife with oxidized lipid molecules, which are generally found in low-density lipoprotein (LDL) molecules, and that are commonly called “bad” fat.
Like a HAZMAT worker entering a contaminated area, the killer T cells responded in the experiments to the hostile world of the tumor microenvironment by increasing their CD36 levels, which drove increased lipid oxidization within itself and resulted in impaired tumor-suppressing powers.
With the basics laid out, they set to work on lab mice to map how CD36 was impairing the anti-cancer functions and found that it did so through a knock-on effect of an increase in a stress response protein.
The good news
The good news is that while the stressed-out T cells couldn’t fight the cancer, a restorative treatment using antibodies to block CD36 production resulted in an increase in glutathione peroxidase, which clears away oxidized lipids, restoring the immune powers.
Glutathione peroxidase is an important enzyme in the regulation of blood lipids, and overweight mice have been shown to have both reduced production of glutathione peroxidase and increased proliferation of prostate cancer cells.
Interestingly in this war of attrition between T cell and tumor, the cure from the tumor’s perspective is also the disease, as too much lipid oxidation also causes tumor cell death, something the researchers noted.
“Now that we’ve uncovered this vulnerability of T cells to lipid oxidation stress, we may need to find more selective approaches to inducing lipid oxidation in the tumor cells but not in the T cells,” said Kaech. “Otherwise, we may destroy the anti-tumor T cells in the process, and our work shows a few interesting possibilities for how to do this”.
Oxidized lipids from our diet come primarily from polyunsaturated fatty acids (PUFAs), and one in particular called linoleic acid, which is the principal PUFA in vegetable and industrial seed oils.
WaL recently reported at length on the oxidized-PUFA hypothesis for heart disease and obesity, and here Salk researchers have added to the potential benefits to be gained from cutting these oils out of our diets.